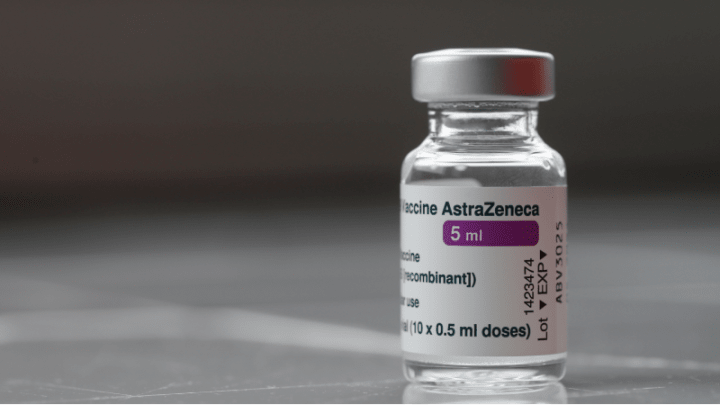
The Oxford-AstraZeneca Covid vaccine is being globally withdrawn due to the pharmaceutical company’s acknowledgment in court documents of a rare and dangerous side effect.
In the European Union, the vaccine is no longer authorized for use as AstraZeneca voluntarily withdrew its marketing authorization. This withdrawal application, submitted on March 5, took effect on Tuesday.
Similar withdrawal applications will be filed in the UK and other countries that previously approved the vaccine, Vaxzevria.
This marks the end of the vaccine’s usage, previously hailed by Boris Johnson as a “triumph for British science” and credited with saving over six million lives.
AstraZeneca clarified that the vaccine is withdrawn from markets for commercial reasons, stating it’s no longer produced or supplied, replaced by updated vaccines targeting new variants.
Vaxzevria faced scrutiny due to a rare side effect causing blood clots and low blood platelet counts. AstraZeneca acknowledged this in court documents filed with the High Court in February, admitting the vaccine “can, in very rare cases, cause TTS.”
Thrombosis with Thrombocytopenia Syndrome (TTS) has been linked to at least 81 deaths in the UK, along with numerous serious injuries. AstraZeneca faces lawsuits from over 50 alleged victims and their families in a High Court case.
Bringing you the latest updates on finance, economies, stocks, bonds, and more. Stay informed with timely insights.